Researchers at Vanderbilt have identified what may be a genetic “smoking gun” for autism spectrum disorder (ASD)—a mutation in the gene for the critical neuronal protein CaMKII. While no single mutation can explain the immensely complicated picture presented by ASD, this study is the first to link a naturally occurring mutation in the CAMK2-alpha gene with a specific neuropsychiatric disorder, said Roger Colbran, Ph.D., professor and interim chair of the Department of Molecular Physiology and Biophysics. Further characterization of the biochemical consequences of this mutation likely will provide novel insights into the molecular mechanisms underlying ASD and may suggest new approaches to improve early diagnosis and/or treatment of the disorder, said Colbran, a Vanderbilt Kennedy Center investigator. The group’s findings were posted online in January by the Journal of Neuroscience.

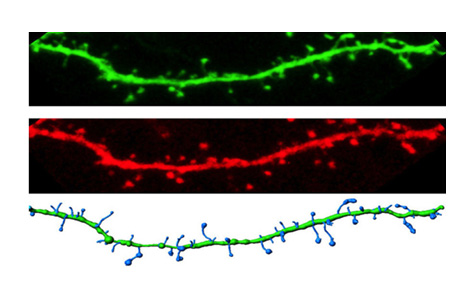
Pictured left to right: Dendrite from a neuron expressing wild type CaMKII. Dendrite from a neuron expressing the ASD-linked E183V mutant CaMKII.
The calcium/calmodulin-dependent protein kinase II (CaMKII) is a key player in synaptic plasticity, learning, and memory. The alpha isoform, encoded by the CAMKIIA gene, is required for long-term potentiation (LTP), the cellular process that leads to the creation of memory. Mice in which CAMKIIA has been “knocked out” exhibit deficits in LTP, learning and memory, increased activity, and aggressive behaviors. Abnormal CaMKII signaling has been linked to several neuropsychiatric disorders.
In the current study, Colbran and colleagues characterized a spontaneously arising CAMKIIA mutation that was identified in a child diagnosed with ASD, and created a transgenic mouse model expressing that mutation to determine how it affected synaptic transmission and behavior. “We show the mutation severely disrupts the biochemical functions of CaMKII in vitro, and induces synaptic abnormalities when the mutant protein is expressed in cultured neurons,” Colbran said. In addition, mice expressing the mutation displayed hyperactivity, deficits in social interaction and increased repetitive behaviors—mimicking the core behavioral features of ASD. The findings suggest that CaMKII plays an important role in the complex molecular pathways that are involved in ASD-related behaviors, the researchers said. It will be important to further analyze the impact of this mutation on synaptic transmission and plasticity in multiple brain regions from these mice. Jason Stephenson, Ph.D., research fellow in Colbran’s lab, was the paper’s first author. Faculty co-authors were Douglas Mortlock, Ph.D., Terunaga Nakagawa, M.D., Ph.D., and James Sutcliffe, Ph.D., a VKC member. The research was supported in part by National Institutes of Health grants MH107765, MH109196, MH063232 and NS078291, and by an intramural Hobbs Discovery Grant from the Vanderbilt Kennedy Center. Bill Snyder is senior science writer, News and Communications, Vanderbilt University Medical Center. Article republished from ResearchNews@Vanderbilt, Feb. 2, 2017.